Cell Line, Process & Product Development
 Home /
Cell Line, Process & Product Development
Home /
Cell Line, Process & Product Development
Transfect genes coding candidate drug products
Apply selection pressure to isolate cells with desired genes
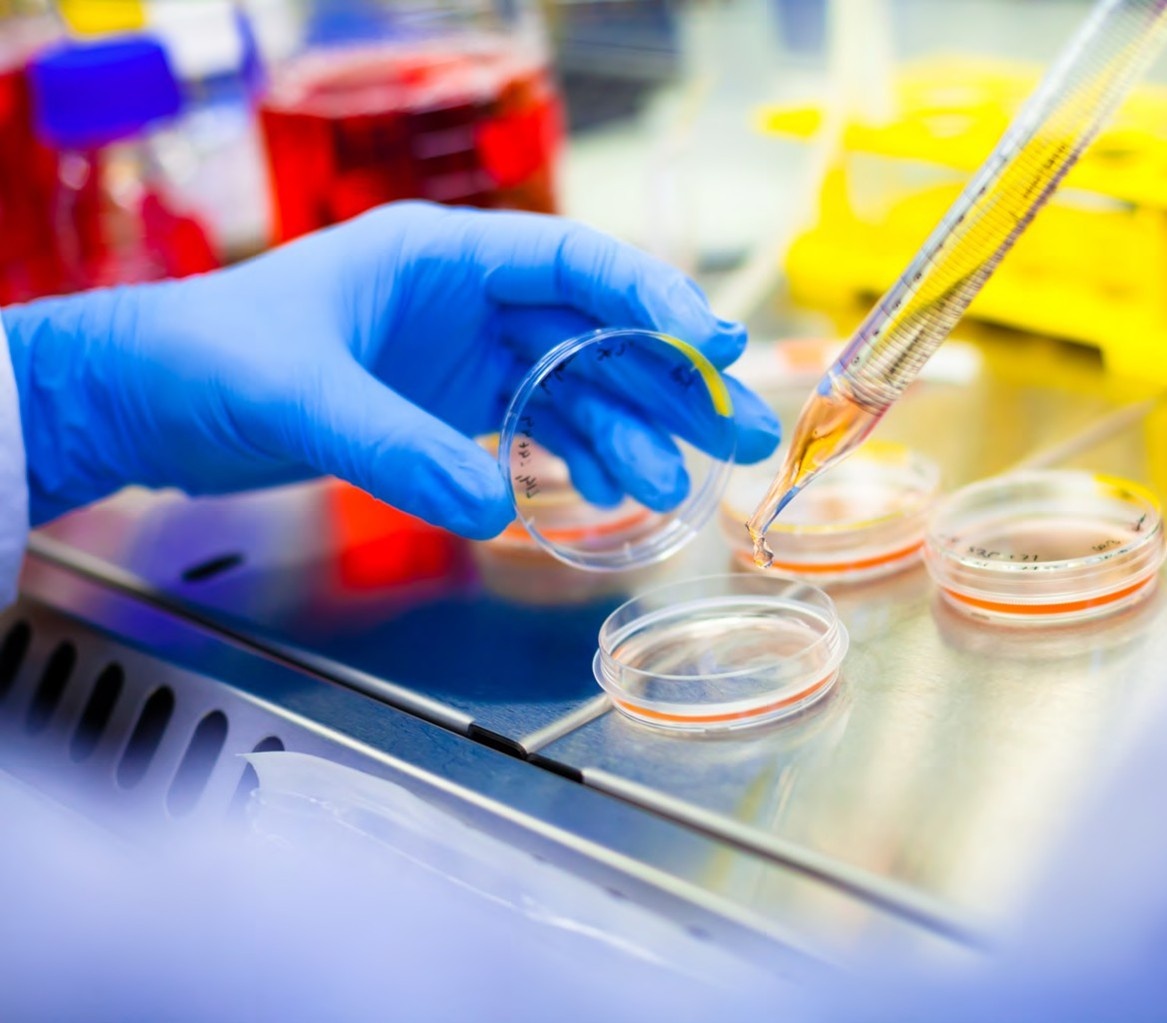
Single cell cloning
High-resolution imaging enables documentation of precise single-cell deposition
Photographic monoclonality evidence supports regulatory compliance
Efficient processes reduce timelines for regulatory approval


Ensures efficient high-yield processes

Ambr bioreactors accelerate process refinement with the aid of Design of Experiment methodologies

XDR-10 & BioBLU enable seamless scale-up

Fine-tune critical parameters for yield, purity, and manufacturability

Smooth transition from lab-scale to clinical production

Optimized workflows speed up therapeutic development
| CQA | Method | Instrument |
|---|---|---|
| Content | Amino Acid Analysis | Agilent 1100 HPLC |
| Copy Number | 7500 FAST qPCR | |
| Function | Binding Kinetics by Biolayer Interferometry | Octet Red384 |
| Structure & Post Translational Modifications | Peptide Mapping | Mass Spec & Agilent 1290 UHPLC |
| Intact Molecular Weight | Mass Spec & Agilent 1290 UHPLC | |
| Subunit Molecular Weight | Mass Spec & Agilent 1290 UHPLC | |
| N-Glycan Profile | Mass Spec & Agilent 1290 UHPLC – FLD |

