GMP Manufacturing
 Home /
GMP Manufacturing
Home /
GMP Manufacturing
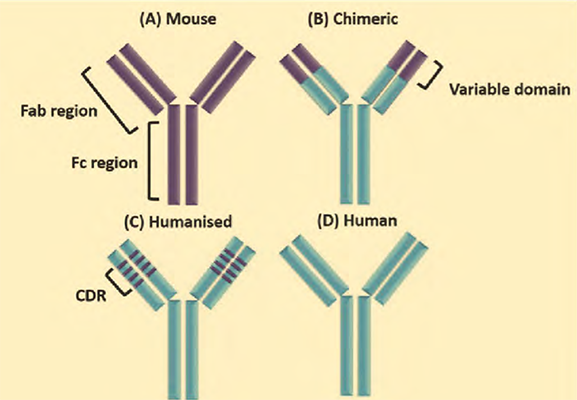

Commercially-proven for manufacturing biologics

CHO (Chinese Hamster Ovary)

HEK-293 (Human Embryonic Kidney)

NS0 (murine myeloma)

Optimized workflows ensure robust biologic manufacturing

Successful development of numerous monoclonal antibody (mAb) drug candidates for a range of indications

Infectious Diseases (Smallpox, Ebola, Marburg)

Autoimmune Disorders (psoriasis, Crohn’s, ulcerative colitis, rheumatoid arthritis).

Proprietary Expression Vector

Demonstrated Scalability and Consistency

Low Manufacturing Cost of Goods

Regulatory — 100% serum-free vs others’ serum dependence

Productivity — Up to 5 gram / L multi-fold higher than SP2/0 or other murine systems

Transfection and Multiplex Selection

The ONLY NS0 platform for single-use manufacturing

Quality Alignment — Produces innovator-specific glycans vs CHO



Mammalian Cell Culture (mAb/glycoprotein production)

200L and 500L Cytiva XDR Bioreactors

Perfusion Intensification (up to 5-fold yield increases)

ÄKTA ready Single-Use Chromatography System

Virus Inactivation and Filtration

Bulk Drug Substance Filling

Fully Single-Use Processes

Easily Transferrable to Large-Scale Manufacturing Sites
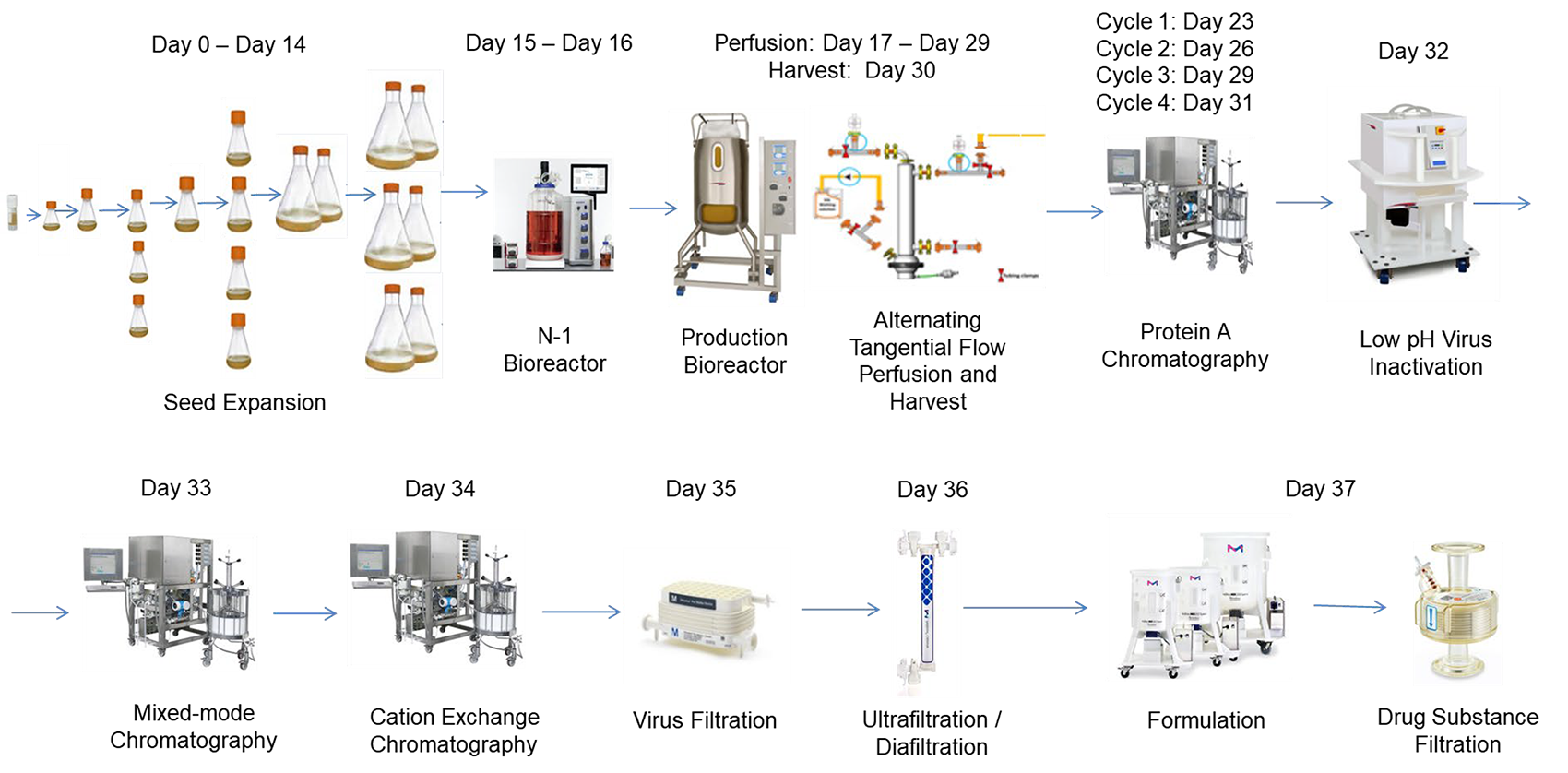

We use Quality by Design (QbD) and Design of Experiments (DOE) to optimize critical process parameters.

Through structured experimentation and data-driven insights, we deliver a high-confidence process design that aligns with regulatory expectations.

We offer perfusion-based validation alongside traditional fed-batch models.

The AMBR® 250 enables efficient DOE execution and it is ideal for accelerating upstream process validation.